Instructors: Taylor Da Silva, Emily Kamma, Fang Fang Li, Josie Setiawan
Lab Technician: Lucia Quesada-Ramirez
Lead: David Ng
Dates: From Oct to June
Grade Level: 11 and 12
Time: 5.5 hours
Start Time: 9am
Running Late? Please call the lab at 604-822-2809
Meeting Spot: Lecture hall in main foyer of the Michael Smith Building (directions)
Cost: Free
In the PCR experiment, each student (and adults) will perform a simplified DNA fingerprint assay where they will check out their own genomic DNA for a genetic insertion. Overall, the lab is pretty fun and easy to do. The students will get a chance to isolate their own DNA, learn about DNA replication, learn about the amazing PCR (polymerase chain reaction) procedure, and they will also run a DNA gel to visualize their results.
The day generally starts at 9:00am or 9:30am and will usually take about 5.5 hours to complete. Your class will have a chance to get lunch during a 1 hour break. In general, any other breaks during the day will be occupied with lecture and are also open season for questions regarding things going on in science or questions regarding science careers and education. We require that at least one adult (the teacher) accompanies the class to facilitate minor supervision (we always have two graduate students on hand), and because the equipment being used is both expensive and delicate we ask that students are told prior to respect the facility. As the material also looks closely at technical considerations of the PCR method, we do ask that all students read this piece on replication, prior to coming to the lab. Note that our instructors are not responsible for disciplining participants in the event of inappropriate behaviour.
Also note that the genetic element characterized is phenotypically null and should carry no ethical consequences (except for the situation where a parent and child elect to do the test together – note that in such a situation, participants will be advised accordingly).
Specifics
DOWNLOAD FIELD TRIP SLIDES (14MB PDF FILE)PCR is an enzymatic amplification process which requires DNA or RNA polymerase, primers and deoxynucleotide triphosphates in an appropriate buffer, and means of controlling the temperature during the various stages of the amplification process. The primers are short single-stranded oligonucleotide sequences complimentary to the 3′ ends flanking the segment of DNA to be amplified. During PCR, the template DNA is denatured to single-strands at a high temperature, then allowed to anneal with an excess of primers at a lower temperature, followed by synthesis of DNA or RNA from the primers at a temperature optimum for the specific polymerase enzyme. After synthesis, the cycle of denaturation, annealing and synthesis is repeated whereby the products of the previous amplification reaction serve as templates for the next round of amplification. This cycle is repeated for as many as 30 times leading to the exponential amplification of the DNA segment of interest. The major product of the PCR amplification process is a double-stranded segment of DNA whose length is defined by the distance between the two primers used and whose termini correspond to the 5′ termini of the primers. The changes in temperature during the cycles can be achieved by using commercially available automated thermal cyclers or by using a series of temperature-controlled water baths. Automated thermocyclers are now in wide in use as they are convenient.
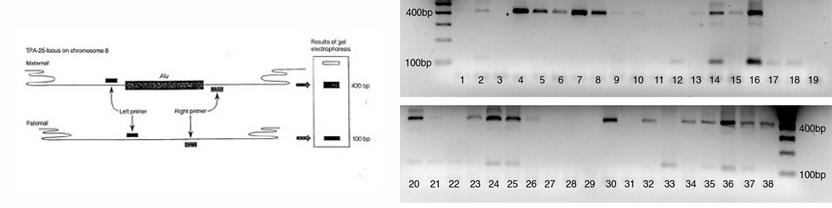
Left: Schematic of PCR reaction. Right: Sample gel showing Alu insertion genotypes
Isolation of DNA from cheek cells:
The source of template DNA for amplification is a sample of several thousand cells obtained by saline mouthwash (bloodless and noninvasive). The cells are collected by centrifugation and resuspended in a solution containing the resin “Chelex. The suspension (containing Chelex) is then boiled to lyse the cells. Following cell lysis Chelex will bind to and remove the proteins and metal ions that inhibit the PCR reaction, but will leave the DNA in the solution. The cell debris and the resin beads are removed by centrifugation. A sample of the supernatant containing chromosomal DNA is mixed with Taq DNA polymerase, oligonucleotide primers, the four deoxyribonucleotides, and the cofactor magnesium chloride. Temperature cycling is used to denature the target DNA, anneal the primers, and extend a complementary DNA strand. The size of the amplification product(s) depends on the presence or absence of the Alu insertion at the TPA-25 locus on each copy of chromosome 8.
Isolate Cheek Cell DNA for PCR Amplification:
1. Obtain 5ml sterile saline capsule, paper cup, and plastic 10ml tube.
2. Remove nib from capsule and squeeze all of the saline solution into your mouth and vigorously swish for 30 seconds.
3. Spit the sample solution into a paper cup.
4. Pour the sample solution from the paper cup into the 10ml tube and close cap tightly.
5. Spin sample in preparatory centrifuge on high speed (1000g) for 10 minutes.
6. Carefully pour off supernatant (liquid on top) into sink and place tube containing your cells on ice.
7. Transfer pellet (using a P1000 micropipettor) to a microcentrifyge tube containing 500ul 10% Chelex solution.
8. Mix cells and Chelex by vortexing vigorously (about 5 seconds) until no visible clumps of cells remain.
9. Place your Chelex (+cheek pellet) tube in a boiling water bath for ten minutes.
12. Carefully remove your tube from the boiling water bath and place on ice for one minute.
13. Place your tube in a microcentrifuge (opposite someone else1s tube!) and centrifuge for 3 minutes.
14. Use a fresh pipet tip to transfer 5 µl of supernatant (clear solution on top – this is your DNA) to a PCR reaction tube.
Detection of an Alu insertion polymorphism by PCR:
Some regions of the human genome show variation between individuals. Such variations in sequence are called polymorphisms and are made to practical use in diagnosis of genetic diseases , forensic identification and paternity testing. In this experiment you would use DNA isolated from your cheek cells (see above) as template to amplify a short region of DNA within the plasmogen activator gene (TPA) containing a short 300bp insertion called an Alu element. Alu elements are short interspersed, repeated DNA elements distributed through out the primate genomes. The Alu element found within an intron of the TPA gene is dimorphic, i.e. it is present in some individuals but not in others. Following PCR amplification and electrophoresis you would be able to determine if the individual is heterozygous or homozygous for the presence or absence of the Alu insertion.
1. Load PCR reaction tube (with your 5ul of supernatent/DNA sample) into a DNA Thermal Cycler and initiate the program specified for the DNA samples in use. Note the following parameters used for this Alu experiment.
PCR MIX:
45ul supermix
1ul Alu Forward primer
1ul Alu Reverse primer
5ul cheek cell DNA sample.
PRIMERS:
Alu Forward 5’-GTAAGAGTTCCGTAACAGGACAGCT-3’
Alu Reverse 5’-CCCCACCCTAGGAGAACTTCTCTTT-3’
PROGRAM (subject to slight changes):
1. 95oC 180 sec (this is the hot start – may not be necessary)
2. 95oC 60sec (denaturation step)
3. 58oC 60sec (annealing of primers step)
4. 72oC 60sec (elongation step)
5. repeat step 2 to 4 (x 45) (45 cycles, enough for a theoretical 3.5 x 1013 fold increase)
Gel Electrophoresis of PCR amplified DNA
7. Prepare a 1% agarose minigel in TBE buffer (with SYBR safe stain).
8. At the end of the PCR reaction, add 10ul of gel loading buffer (blue stuff) to PCR reaction. Load 20ul in gel along with 100bp ladder molecular weight marker.
10. Run the gel for 30min at ~100 Volts constant voltage.
11. Visualize and photograph the gel using Gel Doc system.
12. Analyze the DNA fragments on the gel.
Further Reading and Resources
Here are some links worth checking out.
1. General Background to Molecular Biology
David Secko. A Monk’s Flourishing Garden. The Basics of Molecular Biology Explained (2003) Science Creatively Quarterly
2. Prelude to PCR specifics: You need to get Replication first.
David Ng. Breakfast of Champions Does Replication. (2010) Popperfont.net
3. How does Replication translate to the tweaking of PCR?
Read the “Gist” section on this piece at the Science Creative Quarterly.
Now watch this animation. PCR Animation. From The DNA Learning Center. Assessed November 17th, 2013
4. What to do with your data?
You can actually compare your class’ genotype frequencies with other databases. This one here from ALFRED is an example.